The physical size or shape means nothing in the scheme of things. Electrolytic caps (the cylinders) are layers of an insulative material and HIGHLY etched conductive plates. They etch the plates to make them have greater surface area, which increases the capacitance. This is quite simple, really.
A carbon cap (aka DLC "diamond like carbon" or electrochemical capacitor), functions differently, and far more complexly (is that a word?), closer to the functioning of a battery:
A capacitor is a passive electronic component that stores energy in the electrostatic field that is present at a two-phase interface (for example, solid/liquid or solid/solid). In its simplest form, a capacitor consists of two conducting plates separated by an insulating material called the dielectric. The capacitance is directly proportional to the surface areas of the plates, and is inversely proportional to the plates' separation. In a "double-layer capacitor" (also called an electrochemical capacitor), high-surface-area carbon (~1000 m2/g) serve as the two electrodes which are separated by a liquid electrolyte such as sulfuric acid contained in a thin layer of porous polymer membrane. Because carbon is available in high surface area, and because it is electronically conducting, it is an attractive material for use in capacitors. In addition, carbon is relatively stable in the capacitor environment.
(See image below) When a voltage is applied to the capacitor (and the capacitor is charged), the carbon electrodes attract positive or negative charges (that is, ions that carry the charges) to form double layers at the interfaces. (The figure shows only one electrode and the opposite arrangement of charges exist at the other carbon electrode.). The presence of these charged interfaces is the source of stored electrical energy, which when discharged releases electrons through the external circuit that is connected to the carbon electrodes. In the discharged state, the positive and negative ions are dissipated in the electrolyte and electrode. The major drawback of carbon as an electrode material is its high resistivity because of the poor particle-to-particle contact between the small carbon particles that provide the high surface area. The high resistivity limits the charge/discharge rates. Typical applications include back-up power for small computers and memory devices.
*****IMPORTANT NOTE*****(What this means is that the ESR (equivalent series resistance) will be very high, relative to an electrolytic - when you REALLY want it as low as possible. This means it will not have even CLOSE to the peak current capacity of a good old-fashioned electrolytic capacitor, supposedly the very reason to ADD a cap to your system. Another place where the industry can say "But these go to eleven", and it means nothing for the positive, in fact it can be hindering to the ultimate goal. Numbers are just that - numbers, and can be (and often are) skewed to make you (the consumer) think you are getting more with them than you are with the other guy... --haemphyst)
The distinction between a battery and electrochemical capacitor is not clear-cut. A battery relies on electrochemical reactions that involve active materials in the electrode. In this case, active means that the materials participate in the reaction, and a transfer of electrons between the active material and ionic species in solution occurs across the solid/liquid interface. On the other hand, in an "ideal" electrochemical capacitor that utilizes carbon, the electrodes play a passive role. That is, the electrode surface only participates by serving as sites for charged species to accumulate and no electron transfer occurs across the solid/liquid interface. While in a "practical" electrochemical capacitor there may occur some surface oxidation/reduction on the electrodes, that is, it operates partly as a capacitor, partly as a battery.
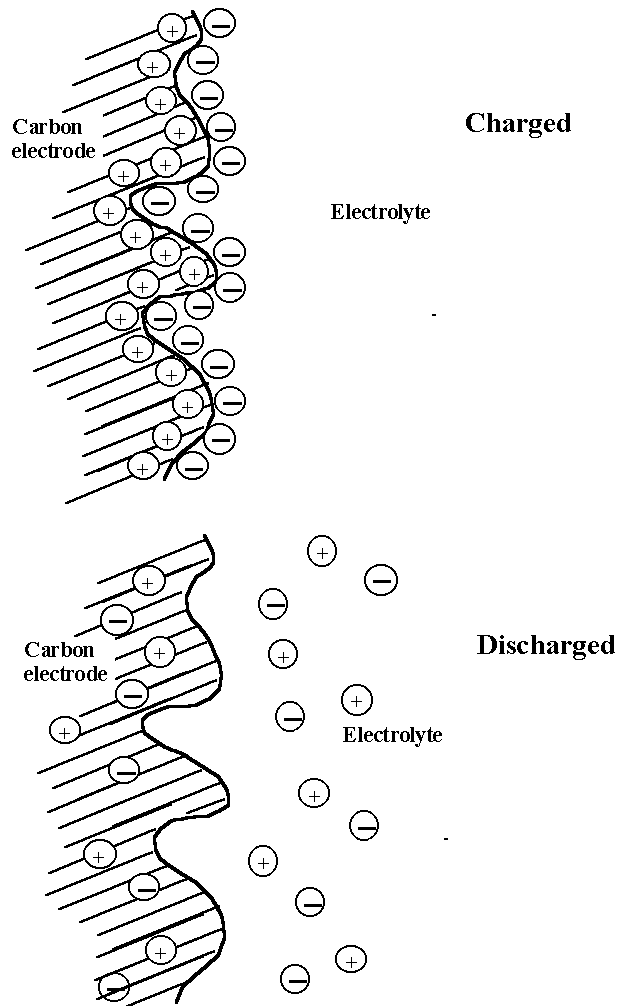
(from Electrochemistry Encyclopedia, found
here.
One more reason to stay with electrolytics? Electrolytic caps will last "forever", so long as they are not subjected to overvoltages, or excessive temperatures. Even if the electrolyte dries up in one, the capacitance will not change dramatically. The carbon cap has a very finite lifespan, also affected by overvoltage conditions and temperature, and like a regular battery, it becomes less effective as it ages.
It all reminds me of something that Molière once said to Guy de Maupassant at a café in Vienna: "That's nice. You should write it down."
 Printable version
Printable version














 (from Electrochemistry Encyclopedia, found
(from Electrochemistry Encyclopedia, found 



